April 25, 2018

The Food and Drug Administration has approved an artificial intelligence device to detect diabetic eye disease, according to reporting in the medical journal Formulary.
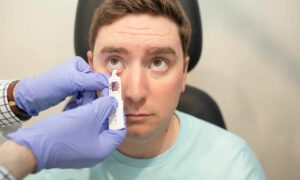
IDx-DR (IDx LLC), a software program that uses an artificial intelligence AI) algorithm to analyze images of the eye taken with a retinal camera, is the first device authorized for marketing that provides a screening decision without the need for a clinician to also interpret the image or results, the FDA said in a statement. That capability, Formulary notes, makes it usable by healthcare providers who may not normally be involved in eyecare.
The way it works is a doctor uploads the digital images of the patient’s retinas to a cloud server on which IDx-DR software is installed. If the images are of sufficient quality, the software provides the doctor with one of two results: (1) “more than mild diabetic retinopathy detected: refer to an eye care professional” or (2) “negative for more than mild diabetic retinopathy; rescreen in 12 months.”
If a positive result is detected, patients should see an eyecare provider for further diagnostic evaluation and possible treatment as soon as possible.